
Careers
How to Choose High Purity Chemicals for Your Laboratory Needs?
Choosing high purity chemicals is crucial for laboratory success. The right chemicals ensure the accuracy of experiments. Impurities can lead to unreliable results. They may alter chemical reactions or produce unexpected outcomes. Reliability in scientific work begins with high purity chemicals.
Consider the intended application of these chemicals. Different experiments require varying levels of purity. Pharmaceutical research demands extremely high purity, while some industrial applications may tolerate more impurities. This raises questions about sourcing. Are all suppliers trustworthy? Do they provide adequate purity information?
Additionally, cost is a practical concern. High purity chemicals often come at a premium price. It’s essential to balance budget with quality. Sometimes, lower-cost chemicals might not be worth the risk. Reflect on what compromises you are willing to accept. Remember, the choice of high purity chemicals directly impacts your research's integrity and success.
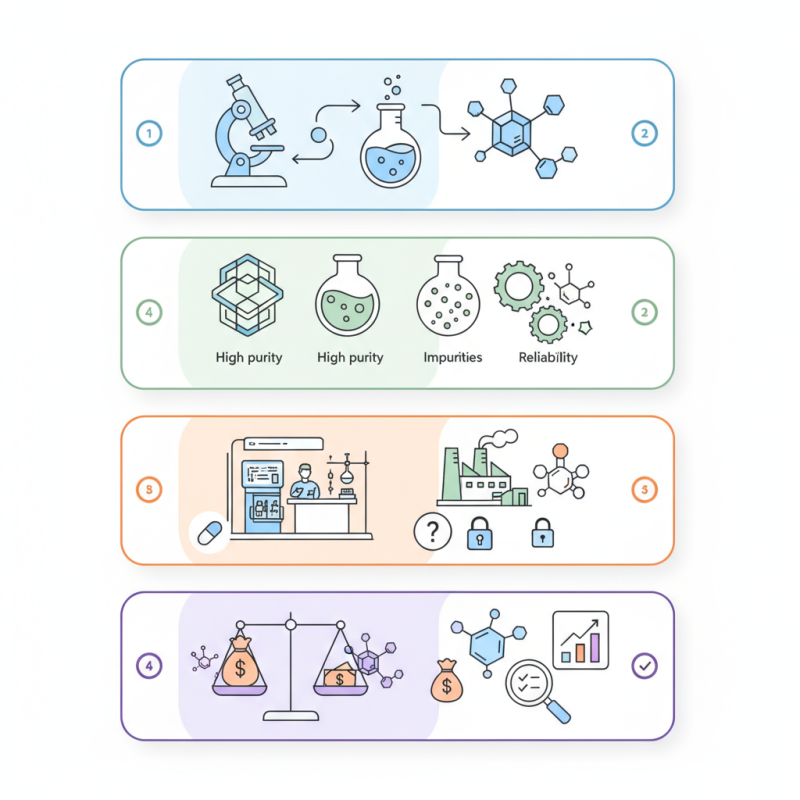
Understanding High Purity Chemicals and Their Importance in Laboratories
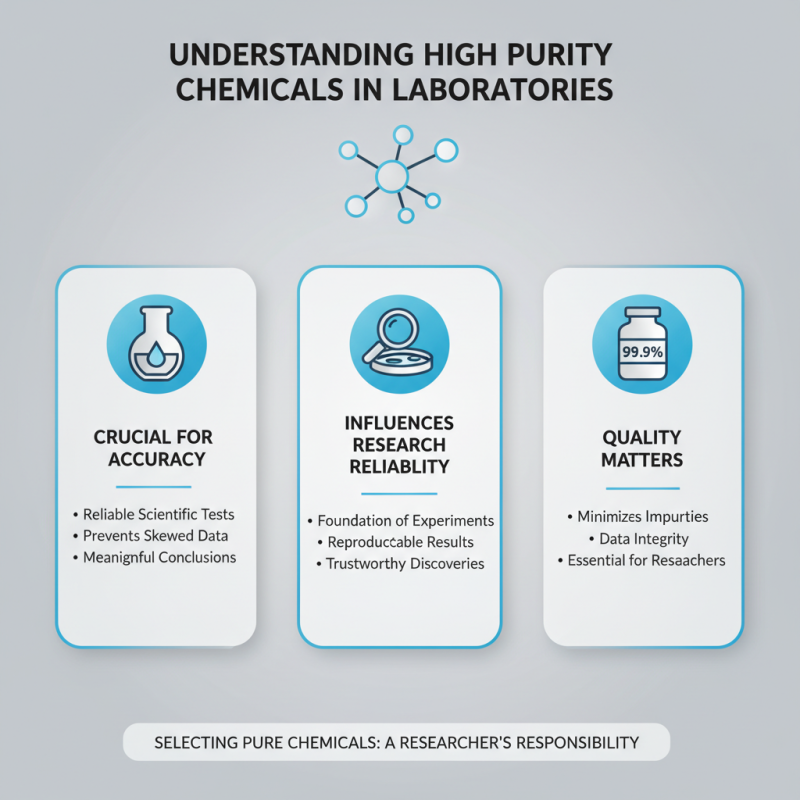
High purity chemicals are vital in laboratory settings. These substances provide accurate results across various scientific tests. Their quality deeply influences the reliability of research and experimentation. Impurities can skew data and lead to misleading conclusions. When selecting chemicals, understanding their purity levels is essential for researchers.
The process of choosing high purity chemicals can be challenging. Many factors must be considered, including solvent composition and potential contaminants. If the wrong chemical is selected, the experiment may yield unusable results. It's not uncommon to overlook minor details in specifications. As scientists, we often focus on broader objectives, but purity should remain in focus. A small impurity could disrupt a significant experiment.
Laboratories often require chemicals with different purity grades. It's tempting to choose higher purity for every task. However, some applications may not require such strict standards. Balancing cost and quality is difficult. Researchers must reflect on their specific needs. Practical considerations sometimes conflict with ideal standards, leading to tough choices.
Key Factors to Consider When Selecting High Purity Chemicals
When selecting high purity chemicals for laboratory use, several key factors must be considered. The purity level is crucial. Generally, chemicals classified with a purity of 99% or higher are deemed suitable for most analytical applications. However, some reports suggest that even minor impurities can significantly affect experimental outcomes. For instance, a study indicated that 0.1% of contamination could alter results in sensitive assays.
The source of chemicals also matters. Buying from reputable suppliers can ensure that the materials meet quality standards. Yet, not all suppliers disclose the methods of purification or testing. This lack of transparency is concerning. Furthermore, using chemicals without verified documentation can lead to unforeseen errors. It’s essential to evaluate the supplier’s quality assurance processes and their history of product reliability.
Storage conditions and expiration dates play a role too. High purity chemicals should be stored properly to avoid degradation. Some compounds can lose their effectiveness even before the expiration date if not handled correctly. Researchers need to be vigilant and maintain proper inventory practices. Regular checks on chemicals can prevent the use of outdated or compromised materials.
How to Choose High Purity Chemicals for Your Laboratory Needs?
| Chemical Name | Purity Level | Application | Storage Conditions | Safety Precautions |
|---|---|---|---|---|
| Acetic Acid | 99.5% | Reagent for organic synthesis | Store in a cool, dry place | Wear gloves and goggles |
| Sodium Chloride | 99.9% | Biological research | Keep in well-closed container | None required |
| Hydrochloric Acid | 37% | pH adjustment | Store in a fume hood | Caustic; wear appropriate PPE |
| Ethanol | 99.5% | Solvent for extraction | Store in a flame-proof cabinet | Highly flammable; avoid open flames |
| Potassium Phosphate | 99.0% | Buffer solutions | Keep dry and sealed | Irritant; use gloves |
Evaluating Suppliers and Sources for Quality Assurance
When evaluating suppliers for high purity chemicals, quality assurance is crucial. Look for suppliers that provide comprehensive Certificates of Analysis (CoA). A CoA details the purity level and the presence of impurities. According to recent industry reports, around 18% of laboratory chemicals fail to meet specified purity standards. This statistic underscores the importance of sourcing from reputable suppliers.
Tip: Always request CoAs for each batch of chemicals. This practice ensures consistency in quality and can prevent experimental errors. Additionally, consider suppliers that participate in third-party quality audits. These audits verify that manufacturers adhere to industry standards.
Another key factor is the supplier’s track record. Investigate their history with other laboratories. Online reviews and case studies can provide insight into their reliability. However, beware of overly positive feedback. Some reviews can be biased or poorly vetted. It’s essential to differentiate genuine feedback from marketing tactics.
Tip: Conduct a comparative analysis of potential suppliers. Look at their safety records, customer support, and complaint resolution efficiency. Making informed decisions based on data and experiences can significantly impact your lab outcomes.
How to Choose High Purity Chemicals for Your Laboratory Needs
This bar chart illustrates the purity levels of high purity chemicals from various suppliers. Ensuring high purity is crucial for laboratory applications, and this comparison can help in evaluating the best sources for chemical procurement.
Common Applications and Uses of High Purity Chemicals in Research

High purity chemicals play a crucial role in modern research. They are essential for accurate experimentation and reliable results. In many cases, even slight impurities can impact experimental outcomes. For instance, in pharmaceutical development, the presence of contaminants can lead to failed drug formulations. According to a recent report, achieving 99.95% purity can significantly enhance the reliability of outcomes in analytical chemistry.
Common applications of high purity chemicals include spectroscopy, chromatography, and synthesis processes. In spectroscopy, high purity solvents help identify compounds without interference. Chromatographic analysis demands precision; therefore, impurities can disrupt results and mislead conclusions. The 2022 Global Chemical Market Report highlighted that around 30% of failed experiments occur due to the use of low-quality reagents.
Laboratories often face challenges when sourcing these chemicals. While high purity is a clear goal, achieving consistent quality can be difficult. Variability among suppliers and batch-to-batch inconsistencies can lead to complications. Researchers should regularly assess supplier quality and perform their purity tests. Ultimately, choosing the right chemicals is a balancing act that impacts research integrity and efficiency.
Best Practices for Storing and Handling High Purity Chemicals
Storing and handling high purity chemicals require careful attention. Proper storage conditions greatly influence the stability and integrity of these substances. According to a 2022 industry report, improper storage can lead to contamination or degradation, affecting experimental outcomes. It recommends a controlled environment, with specific temperature and humidity levels for different chemicals.
Containers must be correctly labeled. Mislabeling can cause significant errors. Industry data indicates that nearly 30% of lab accidents stem from handling mishaps. Clear, accurate labels help minimize such risks. Additionally, using the right container material is crucial. Glass, for example, can be suitable for many substances, but some high purity chemicals require specialized materials to prevent reactions.
When handling, it's crucial to employ proper personal protective equipment. This minimizes risks for laboratory personnel. A report from the Occupational Safety and Health Administration noted that inadequate protective measures resulted in increased workplace injuries. Employees must be trained regularly to recognize hazards associated with these chemicals. However, many labs overlook this vital aspect, leading to potential disasters. Regular audits and refresher courses can bridge this gap, fostering a safer laboratory environment.
Related Posts
-
Top High Purity Chemicals for Advanced Applications and Their Benefits?
-
How to Choose the Right Manufacturing Chemical for Your Needs?
-

How to Choose and Use High Purity Chemicals Effectively?
-

How to Get Ultra Pure Chemicals: A Step-by-Step Guide for Beginners
-

10 Best Industrial Chemical Solutions for Your Business Needs?
-

Top 5 Chemical Analysis Techniques Every Scientist Should Know


